SEMICONDUCTOR TECHNOLOGY Scientists develop the newest semiconductor of 2024
Related Vendors
Scientists at Georgia Institute of Technology in Atlanta, USA claim that they have transformed the zero band gap material “Graphene” into a fully operational semiconductor. SEG: Semiconducting Epitaxial Graphene is said to be robust and faster than traditional silicon-based nanoelectronics. This article explains graphene’s original metallic properties and the advancement of band gap engineering to make it a semiconductor.

Suppose you take sand in your hands while someone tells you that these tiny grains become the chip in your laptop. That’s how fabrication unit “fabs” extract silicon from sand to chemically process it for manufacturing semiconductor chips. Imagine again that once you break the tip of your sharp pencil, a grayish non-sticky material “graphite” leaves a mark on your hand. Another similar material “Graphene” will now create semiconductor chips that empower our phones, laptops, and many devices.
Graphene has always been a subject of interest for scientists due to its excellent electrical conductivity, transmittance, physical properties, and availability. Months after Germany’s Fraunhofer IISB introduced the Aluminium Nitride wafer, the industry now talks about graphene-based semiconductors.
The metallic block of graphene
Graphene is an allotrope of carbon. In simple words, graphene's internal structure comprises carbon atoms arranged in a hexagonal lattice to form sigma and pi covalent bonds. The electronic configuration of carbon is 1s2 2s2 2p2. Each carbon electron in orbitals 2s, 2px, and 2py forms three sigma bonds with the nearest neighboring atoms. The overlapping of orbitals 2s, 2px, and 2py results in the creation of an sp2 hybrid orbital.
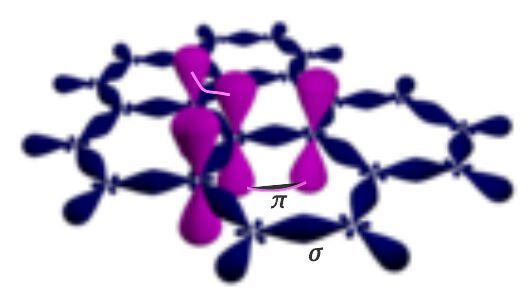
In addition, tunneling in 2pz orbitals form pi bonds that contribute to the delocalized pi system. Six electrons contribute to the sigma bond and two form pi bonds. The sigma bonds are the strongest covalent bonds compared to pi bonds. The number of electrons involved in a pi bond is called a “delocalized electron”. The delocalized electrons have high energy and are free to move between valence and conduction bands.
:quality(80):fill(efefef,0)/p7i.vogel.de/wcms/65/40/6540aa7ec3a0b/whitepaper-littelfuse-coverbild.png)
Originally not a semiconductor
It is a fact that semiconductor characteristics are introduced through band gaps. However, graphene has a zero band gap. The absence of an energy gap between the valence and conduction band makes graphene an excellent conductor of electricity. Even in undoped graphene, valence, and conduction bands form a Dirac cone. To unlock graphene's band gap, researchers at Georgia Institute of Technology in Atlanta, USA claim to have engineered a material called “SEG” under a process called quasi-equilibrium annealing.
A scientific effort to make graphene a semiconductor
The theoretical road
The attempts to make graphene a semiconductor were discussed only on paper for a decade. There was no practical structure that could demonstrate the semiconductive behavior of graphene. The initial consideration was to create graphene nanoribbons. A study published in 2022, talked about engineering graphene under hydrostatic conditions. The proposed pressure is between 1 to 30 GPa with the gases boron nitride and molybdenum dioxide to form a superlattice. The high pressure modifies the inter-lattice distance for introducing a band gap in the material. However, the practical results did not exhibit semiconductive character.
:quality(80):fill(efefef,0)/p7i.vogel.de/wcms/5f/fe/5ffedb2e0ffa6/listing.jpg)
Practical success
The Graphene-based semiconductor is called “Semiconductor Epitaxial Graphene” (SEG). SEG is an ordered buffer layer grown over a SiC crystal to achieve an aligned lattice. The chemical process extends the band gap of the 2D semiconductor up to 0.6 V, almost equal to silicon. The report also claims that a prototype SEG FET exhibits ambipolar diffusion and fine-switching characteristics.
Scientists observed that the method to separate silicon from silicon carbide substrate forms a disordered insulating layer of graphene. The new annealing method heats the compound to 1000 degrees and cools it down to achieve the desirable material. The resulting covalent bonds are strong and ordered. The delocalized pi electrons are mobile at higher speeds to increase the band gap.
Epigraphene is an ordered buffer layer grown over SiC crystal after eliminating silicon. Advanced microscopic techniques distinguish between SiC and epi graphene during the procedure. The device has a high carrier density at room temperatures larger than commercially available semiconductors to enable terahertz frequency operation. The scientist behind the invention- Walter De Heer quotes “Graphene can take over and be the paradigm for the next 50 years.
What's next for SEG?
Summarizing properties of semiconducting epitaxial graphene “SEG”:
- Chemically and mechanically robust
- High room temperature mobility
- 10x faster than silicon semiconductors
- Ambipolar diffusion
- Lesser heat dissipation
- Reasonable IV characteristics
- Abundance in nature
- Compatible with all standard fabrication processes
The practical existence of Moore’s law is pressuring the industry to achieve miniaturization. Decreasing the chip size further can lead to the CPU bottleneck. Industry focuses on introducing new semiconductors to reduce reliance on silicon, and fabrication costs while improving efficiency. The researchers behind the technological achievement compare the invention of SEG to silicon chips. De Heer comments “Thinking silicon is the end of electronics, it’s not. It’s the beginning”. SEG is said to be more efficient, abundant, and faster than silicon semiconductors. The possible applications could be power electronics, deep space, RF, 6G, and quantum computing. However, scaling SEG production to the level of silicon-based semiconductors with a low fabrication cost remains a challenge in the hardware industry.
(ID:49883860)



:quality(80)/p7i.vogel.de/wcms/2f/f3/2ff3221bf7665de2d0acf83760bfd1fa/0130031523v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c3/16/c316e955a97f5d72d9678297b237b9e5/0129932858v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/ef/0a/ef0adb0acf793fe147cc27c21f6a7a67/0129954238v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/53/f9/53f9301dfc9292d02960f7996c79cc6e/0129927601v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6e/cd/6ecd41d095d5111cf4ed37b714844487/0129930878v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/02/c0/02c0e9722f70b1134dbf96fb59a9c73d/0129655179v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/cc/67/cc670ea2029cd2af5c641af70e1bf734/0129816392v4.jpeg)
:quality(80)/p7i.vogel.de/wcms/ea/e6/eae6aee30071e67a5627027974437134/0129544613v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/23/1a/231a97643aa7fc3adf2704a6273756c3/0130030642v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3c/4b/3c4bbc68dda65c7071b4376fb57423b0/0130030465v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b1/5e/b15ee02b0ba02db70cf61e37d66ad1d3/0129349127v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/26/d5/26d591cc340077026eac56a0e7564faf/0129949603v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/c8/bec8d43fc0ee73414274be44608b2970/0129748903v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/23/ee/23ee4a97790d6009dbfd7d9577ffa723/0129220424v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3c/d1/3cd1cacbceb792ba63727199c61ca434/0127801860v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/5a/a0/5aa0436498af618297961fd54ab36cdf/0126290792v2.jpeg)
:fill(fff,0)/p7i.vogel.de/companies/60/7e/607ec89d5d9b5/white-frame.jpg)
:fill(fff,0)/p7i.vogel.de/companies/68/00/6800eb0040fb9/cobalt-vertical-aligned-bold.png)
:fill(fff,0)/p7i.vogel.de/companies/62/95/6295c25c8dc1a/schunk-sonosystems-300dpi.png)
:quality(80)/p7i.vogel.de/wcms/0c/71/0c7136f0406689ac88a83b3d5c9404c8/0123816960v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/7d/40/7d406bd959b3a9127c33a66157f9030a/0128339184v4.jpeg)