BANDGAP ENERGY Direct vs indirect bandgap semiconductors
Related Vendors
Direct vs indirect bandgap semiconductors differ in the alignment of energy bands, making recombination of electrons and holes different. The photonics and renewable energy industries look forward to direct-bandgap semiconductors, while standard electronics is built on indirect-bandgap semiconductors. The power industry relies on both materials. This article explains direct vs indirect bandgap semiconductors, shares a list categorizing industrial semiconductors, and explains why engineers should understand the difference between the two.

Direct bandgap semiconductors emit light, whereas indirect bandgap semiconductors release heat. In direct bandgap semiconductors like gallium arsenide (1.42 eV), the valence and conduction bands are aligned with respect to the momentum. Electron-hole recombination readily releases light (photons). Direct bandgap semiconductors are used in LEDs, laser diodes, and solar cells. In indirect bandgap semiconductors, including silicon (1.12 eV) and silicon carbide (3.26 eV), the valence band and conduction band are misaligned at different values with respect to the momentum. The recombination releases heat instead of light. Direct vs indirect bandgap semiconductors can indicate whether a material is suitable for a laser or a transistor.
Why should power engineers choose between direct and indirect bandgap semiconductors?
The poor performance of silicon in high-power electronics has always driven engineers to seek alternatives. The advent of wide bandgap semiconductors is driven by silicon carbide (SiC) and gallium nitride (GaN). Surprisingly, SiC and GaN sit on different ends of the direct vs indirect bandgap spectrum. SiC is an indirect bandgap semiconductor, while GaN is a direct bandgap semiconductor.
Whether engineers should choose between SiC or GaN depends on properties such as high breakdown field, power density, electron mobility, and thermal conductivity. Direct and indirect bandgap behavior doesn’t affect the choice. The distinction between direct and indirect bandgap semiconductors is critical for any application that needs light-to-electricity conversion, or vice versa.
When light strikes the semiconductor surface, it doesn’t get absorbed during the first step, but enters some distance into the material. Absorption coefficient is a metric that indicates light absorption over a short distance. It is expressed per cm or per micrometer.
Direct bandgap example semiconductors exhibit a high absorption coefficient near the band edge. Indirect bandgap semiconductors exhibit a low absorption coefficient near the band edge. When the photonic energy exceeds the bandgap, the absorption coefficient becomes large in direct bandgap semiconductors. In indirect bandgap semiconductors, the absorption coefficient increases gradually, but remains low with the participation of phonons.
The absorption coefficient matters if foundries manufacture and design solar cells for home and commercial use. Engineers use optical absorption spectroscopy to measure band gap energy, along with the Tauc plot of absorption coefficient vs photon energy, for analysis. Manufacturers can use the direct GaN bandgap on the die to make photonic devices.
Silicon-based PV modules (Indirect bandgap example) are manufactured using thick wafers. These modules are larger and found on rooftops. GaAs bandgap focuses on low material cost per area. Perovskite solar cells are thinner and consume tiny amounts of power per watt. Most photonic devices, including photodetectors, are made from other compounds in the III-V group rather than silicon.
:quality(80):fill(efefef,0)/p7i.vogel.de/wcms/6a/02/6a02d06fa2e86/wp-magnachip-sustainably-strong--mastering-the-reduction-of-oxides-coverbild.jpeg)
Direct vs indirect materials at a glance
Silicon and germanium, the most common semiconductors, exhibit an indirect bandgap. Most group III-V compound semiconductors have indirect band gaps. It is interesting to note that boron nitride, an insulator used in the semiconductor industry, behaves as a switch between direct and indirect band gaps under different operating conditions. The table lists examples of direct and indirect bandgap materials.
| Semiconductor material name | Bandgap type | Bandgap energy at 300 K (Room temperature) | Applications |
| Silicon (Si) | Indirect bandgap | 1.12 eV | Standard electronics |
| Germanium (Ge) | Indirect bandgap | 0.7 eV | Historic semiconductor used in Schottky diodes, rock musician transistors, and radar pulse detection. Current applications with silicon (SiGe BiCMOS) in fiber optics, infrared components, imaging cameras, IR detection astronomy, and RF components |
| Silicon carbide (4H-SiC) | Indirect bandgap | 3.26 eV | EV traction, railway traction, solar inverters, industrial motor drive, power supplies, and three-phase converters |
| Gallium phosphide (GaP) | Indirect bandgap | 2.26 eV | Green/Red LEDs with nitrogen-doped engineering |
| Gallium oxide (-Ga2O3) | Indirect bandgap | 4.8 eV | Deep UV detection (DUV), solar-blind UV photodetectors, tunable devices in RF, and potential for multi-kV JFETs and MOSFETs, and are actively researched by enterprises to manufacture 6-inch wafers |
| Diamond | Indirect bandgap | 5.47 eV | Heat sinks and spreaders in power electronics, RF, superconductors, and actively researched to engineer power transistors and power diodes |
| Gallium arsenide (GaAs) | Direct bandgap | 1.42 eV | Infrared LEDs, laser diodes, solar cells, and microwave applications |
| Gallium nitride (GaN) | Direct bandgap | 3.4 eV | Blue/white LEDs, laser diodes, HEMT for fast charging, data center power, RF amplifier, consumer electronics like GaN charger, multi-level converters, HVAC |
| Indium phosphide (InP) | Direct bandgap | 1.34 eV | Telecom lasers, yellow/orange LEDs, and photodetectors |
| Aluminum nitride (AlN) | Direct bandgap | 6.2 eV | Silicon-on-insulator, emerging 100-mm AlN wafers applicable in power electronics and RF devices, optical storage media, microwave packaging, substrates in optoelectronics, ultra-wide bandgap power electronic substrates |
| Boron nitride (BN) | Indirect and direct bandgap both | 5.8 eV | Laser printers, thermal management materials, lubricants, nanotubes, insulation in semiconductor fabrication processes, and as a potential semiconductor in energy storage due to bandgap switching properties |
How does the direct-indirect bandgap distinction work?
Bandgap, also known as the forbidden gap, is a small distance between the conduction and valence bands. The valence band exhibits the lowest energy and contains all the stable electrons. The conduction band is empty and exhibits the highest energy. By applying a significant amount of energy, electrons can jump from low-energy valence bands to high-energy conduction bands.
The amount of energy required to move an electron between both bands is measurable in electron volts (eV). It is known as the band gap energy. Every electron inside a crystal is defined by its energy and crystal momentum “k-vector”. As per the law of physics, both energy and momentum must be conserved during any reaction.
:quality(80)/p7i.vogel.de/wcms/f0/a1/f0a1ae7496e69c9ec84186d7ebdf897d/0130870335v2.jpeg)
SEMICONDUCTORS
The material footprint of the energy transition
When an electron drops from the conduction band to the valence band, it is said to recombine with the hole in the valence band. As per the law of conservation of energy, the recombination energy is released. Momentum is also conserved. Band alignment, recombination behavior, and momentum transfer decide whether a material is a direct vs indirect bandgap semiconductor.
The band gap energy diagram, known as the E-k diagram, plots momentum (k-vector) on the horizontal X-axis and energy on the vertical Y-axis. Valence and conduction bands are shown as curved regions.
Direct bandgap semiconductors: Direct bandgap (DBG) semiconductors have aligned conduction and valence bands at the same momentum k-values.
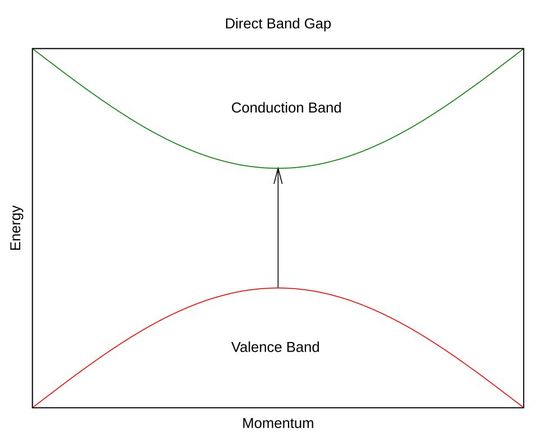
In direct bandgap semiconductors, the minimum of the conduction band and the maximum of the valence band occur at the same crystal momentum k-vector in the allowable states known as the Brillouin zone.
Electrons can easily jump from the conduction band to recombine with holes in the valence band. The extra energy is released as photons. These photons carry energy, but exhibit zero momentum, which doesn’t matter inside the crystal.
Indirect bandgap semiconductors:
Indirect bandgap (IBG) semiconductors are materials in which the minimum of the conduction band and the maximum of the valence band occur at different values of crystal momentum k-vectors in the Brillouin zone.
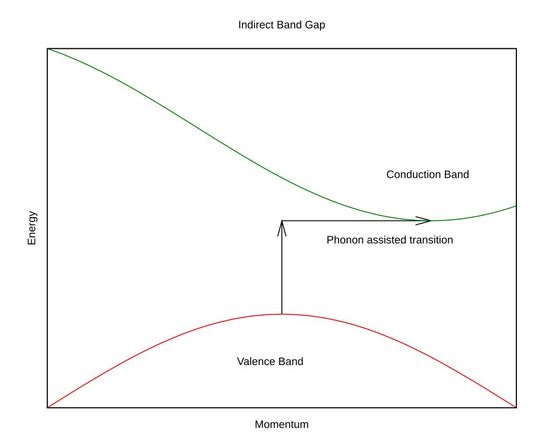
As photons don’t exhibit enough momentum, a lattice vibration known as a phonon supplies the missing momentum. The phonon is either absorbed or emitted during the recombination process.
The interaction between electrons, photons, and phonons in indirect bandgap semiconductors is known as the three-body interaction. Three particles cannot coordinate quantum-mechanically, making the transition probability extremely low, about 1 in a million.
As a consequence, indirect bandgap semiconductors cannot emit light like direct bandgap semiconductors. Indirect bandgap semiconductors like silicon release heat during operation. The radiative lifetime of silicon is 1 millisecond, a million times slower than gallium arsenide. Indirect bandgap examples include silicon and silicon carbide.
-
Feature
Direct bandgap semiconductors
Indirect bandgap semiconductors
Band alignment
The maximum of the valence band and the minimum of the conduction band exist at the same momentum k-value
The maximum of the valence band and the minimum of the conduction band exist at different momentum k-values
Recombination result
Radiative, energy released as light
Non-radiative, energy released as heat
Photon release
Easy, single-step
Tough, three-step assisted by phonon
Absorption coefficient near the band edge
High
Low
Applications
Optoelectronics, photonics, and power electronics
Standard electronics
References
- https://www.doitpoms.ac.uk/tlplib/semiconductors/direct.php
- https://www.sciencedirect.com/topics/engineering/indirect-semiconductor
- https://www.researchgate.net/figure/Absorption-coefficient-of-some-direct-and-indirect-gap-semiconductors-For-the-direct-gap_fig2_268059459
(ID:50846749)



:quality(80)/p7i.vogel.de/wcms/e1/fc/e1fc24c34815d25f3b025993efe2291e/0131176746v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/b8/d3/b8d3ec2126c031b4b96aa350228ae357/0131175054v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/55/5b/555bb8759d60243792dd736b86585f29/0130909253v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/db/75/db7507346cd4842ca5eb8838f45861ce/0130908924v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/21/82/21825475e21f7e3e77cffb9c2665c5e1/0131273557v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/97/c0/97c0f847c457054bfb41a3388dd9fc4c/0131184610v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/71/74/71744e9858925f4aae140ab39723ebf4/0131064963v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/8a/2a/8a2a43ba86d8fbb6089aa249a066e1f5/0131041579v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/63/d5/63d5cb8e8029d9826e610bd1c54a00ad/0131310853v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3d/d0/3dd068b74bc92e76f00a3051fc28558b/0131309601v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/0a/fa/0afa82d35905525e2eada79d976c3b53/0130924910v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/fb/d4/fbd46e4d264fb476772111e184e23aa1/0131187942v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/34/4a/344a3dc5a19429cf6726b7f8f7e2ee32/0130109719v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/c8/bec8d43fc0ee73414274be44608b2970/0129748903v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/23/ee/23ee4a97790d6009dbfd7d9577ffa723/0129220424v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/3c/d1/3cd1cacbceb792ba63727199c61ca434/0127801860v2.jpeg)
:fill(fff,0)/p7i.vogel.de/companies/63/c7/63c7da97be945/diotec.png)
:fill(fff,0)/p7i.vogel.de/companies/66/8b/668becd1c07eb/dowa-logo-word--1-.jpeg)
:fill(fff,0)/p7i.vogel.de/companies/68/08/6808a2b3b6595/het-logo.jpeg)
:quality(80)/p7i.vogel.de/wcms/7b/2c/7b2c4ddd6c8fd3b44fe8afc18f0d7e40/0115676667.jpeg)
:quality(80)/p7i.vogel.de/wcms/e6/49/e6495c2d66253b8a488f774bed717d0b/0130452223v2.jpeg)